Which Best Demonstrates an Example of the Common Ion Effect
Which best demonstrates an example of the common ion effect. There is no best example because any one prokaryotic cell is equally prokaryotic as the next.

Interaction Based Ion Selectivity Exhibited By Self Assembled Cross Linked Zwitterionic Copolymer Membranes Pnas
HCl H Cl.

. Ammonia is produced commercially when an increase in pressure shifts the equilibrium toward the product. ту B Ammonia is produced commercially when an increase in pressure shifts the equilibrium toward the product. The reduction of the degree of dissociation of a salt by the addition of a common-ion is called the common ion effect.
What is the empirical formula of this compound. When sodium chloride is added to. This makes H a common ion and creates a common ion effect.
10799999997667 x 10 11. Which best demonstrates an example of the common ion effect. The common ion effect is a decrease in the solubility of an ionic compound as a result of the addition of a common ion.
This is called common Ion effect. The most common example would probably be E. Which best demonstrates an example of the common ion effect The compound CuCl is 500 times less soluble in sea water than it is in pure water.
A compound consists of 347 grams of oxygen and 152 grams of nitrogen. Harrison pours sand into a bucket at a constant rate. 54 x 10 13 0050 s s The produces a quadratic.
Adding calcium ion to the saturated solution of calcium sulfate causes additional CaSO 4 to precipitate from the solution lowering its solubility. The addition of a solution containing sulfate ion such as. The common-ion effect is an example of chemical equilibrium.
Acetic acid being a weak acid ionizes to a small extent as. After 10 seconds the total weight is 50 ounces. Common-ion effect decrease in solubility of an ionic salt ie one that dissociates in solution into its ions caused by the presence in solution of another solute that contains one of the same ions as the salt.
In a saturated solution of silver chloride we have the equilibrium. Thus the ionization of H 2 S is decreased. Ompounds C A lighted splint goes out in air but bursts into flame when plunged into.
Duction A The compound CuCl is 500 times less soluble in sea water than it is in pure water. For example silver chloride AgCl is a slightly soluble salt that in solution dissociates into the ions Ag and Cl -. Common-ion effect decrease in solubility of an ionic salt ie one that dissociates in solution into its ions caused by the presence in solution of another solute that contains one of the same ions as the salt.
The concentration of un-ionized H 2 S is increased. Due to the increase in concentration of H ions the equilibrium of dissociation of H 2 S shifts to the left and keeps the value of K a constant. In the above example the common ion is Ca 2.
The common-ion effect is an example of chemical equilibrium. What is an equation of the line parallel to the line on the graph that passes through 225. The graph shows the total weight of the bucket and the sand over time.
S 2 005s - 54 x 10 13 0 Substituting into a quadratic solver gives. Ag C l. Which best demonstrates an example of the common ion effect.
The decomposition of carbonic acid in water occurs spontaneously whereas the combination of carbon dioxide and water does not. Duction A The compound CuCl is 500 times less soluble in sea water than it is in. In other words the ignoring was a perfectly valid thing to do.
CH3COOH CH3COO H. If to an ionic equilibrium AB A B a salt containing a common ion is added the equilibrium shifts in the backward direction. If you decided that the additional amount of silver ion CANNOT be ignored you would have this.
THIS SET IS OFTEN IN FOLDERS WITH. To this solution suppose the salt of this weak acid with a strong base is added.

17 4 Solubility Of Salts Chemistry Libretexts

9 Ionic Bond Examples In Daily Life Studiousguy

Applications Of Ionic Liquids In The Chemical Industry Chemical Society Reviews Rsc Publishing Doi 10 1039 B006677j

Buffer Capacity Video Buffer Solutions Khan Academy

9 Ionic Bond Examples In Daily Life Studiousguy
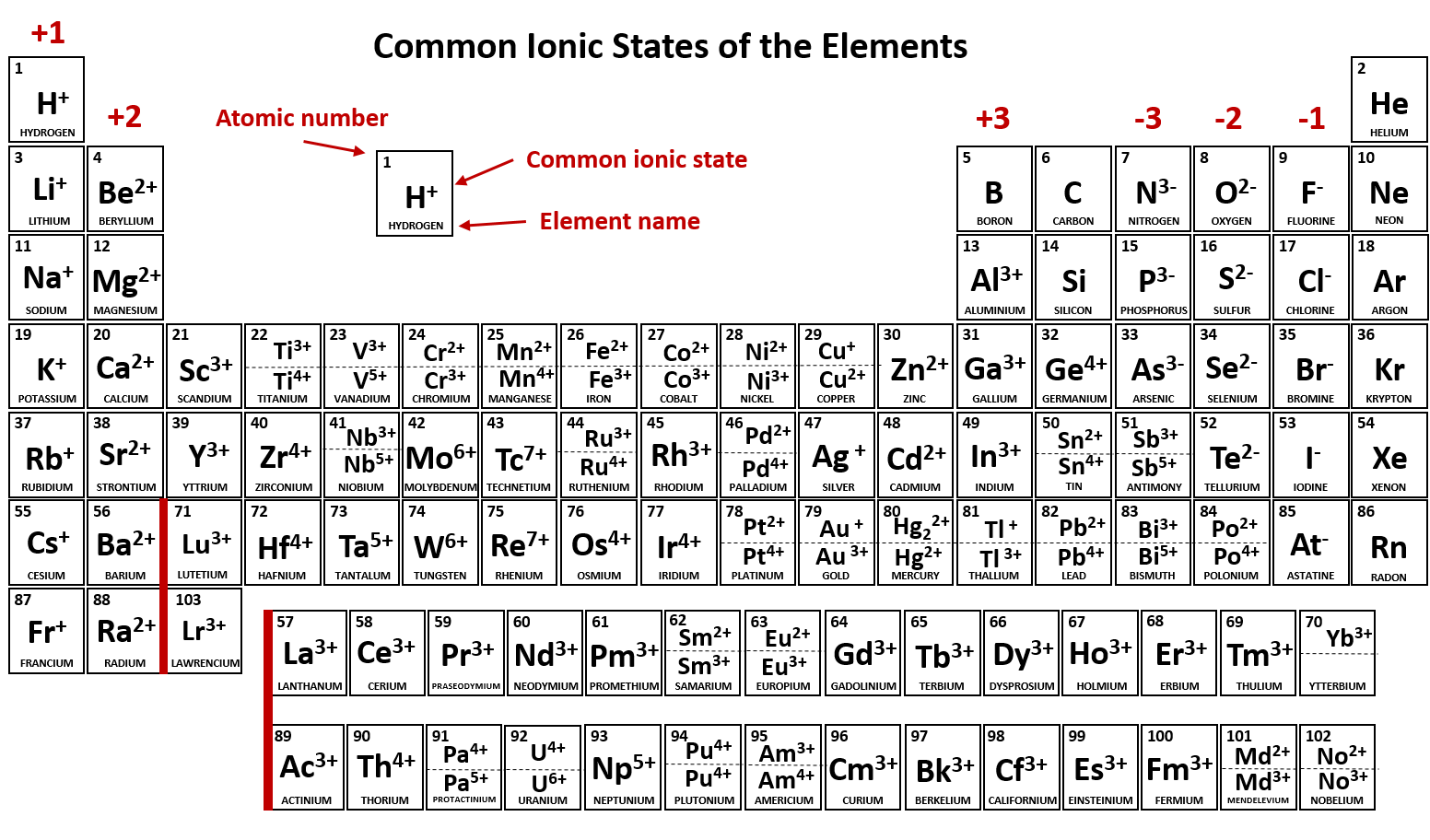
Ch104 Chapter 3 Ions And Ionic Compounds Chemistry

Solubility Properties Of Ionic Compounds Chemistry Jove
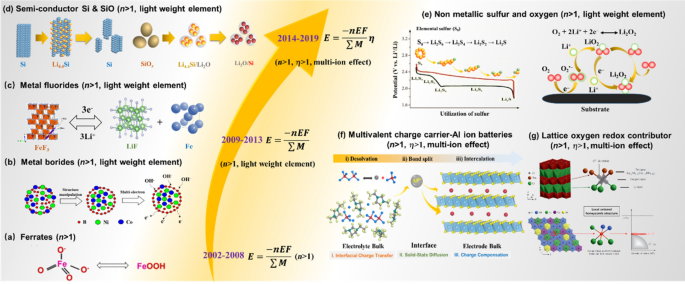
Multi Electron Reaction Materials For High Energy Density Secondary Batteries Current Status And Prospective Springerlink

Calcium Ion An Overview Sciencedirect Topics

17 4 Solubility Of Salts Chemistry Libretexts

Ion Permeability An Overview Sciencedirect Topics

Recent Progress Of Task Specific Ionic Liquids In Chiral Resolution And Extraction Of Biological Samples And Metal Ions Wu 2018 Journal Of Separation Science Wiley Online Library

18 3 Common Ion Effect In Solubility Equilibria Chemistry Libretexts

Pdf Production Of Biodiesel Using Ionic Liquids
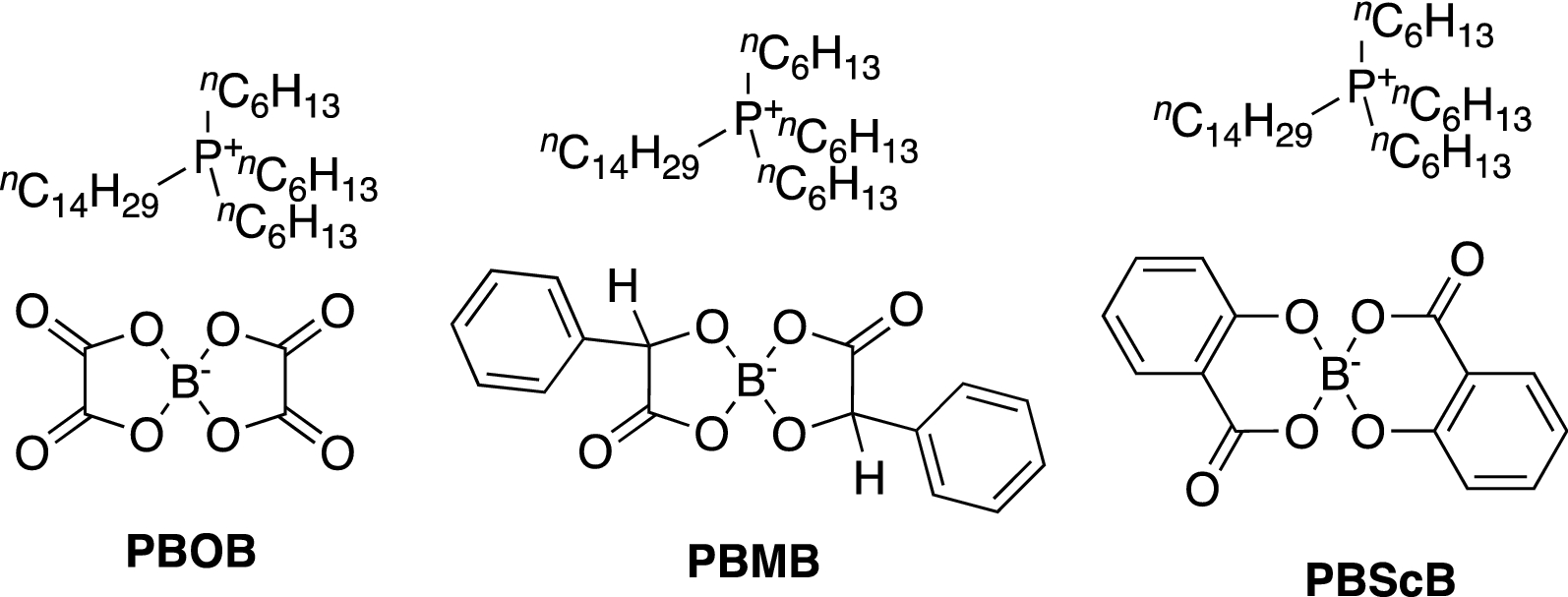
The Effect Of Anion Architecture On The Lubrication Chemistry Of Phosphonium Orthoborate Ionic Liquids Scientific Reports

Chemistry The Central Science Chapter 17 Section 5



Comments
Post a Comment